Quality Assurance & Quality Control In Pharma Industry – ppt download
Presentation on theme: “Quality Assurance & Quality Control In Pharma Industry”— Presentation transcript:
1
Quality Assurance & Quality Control In Pharma Industry
Dr. Basavaraj K. Nanjwade M.Pharm., Ph. D Associate Professor of Pharmaceutics Department of Pharmaceutics KLE University, Belgaum – , Karnataka, INDIA Cell No:

2
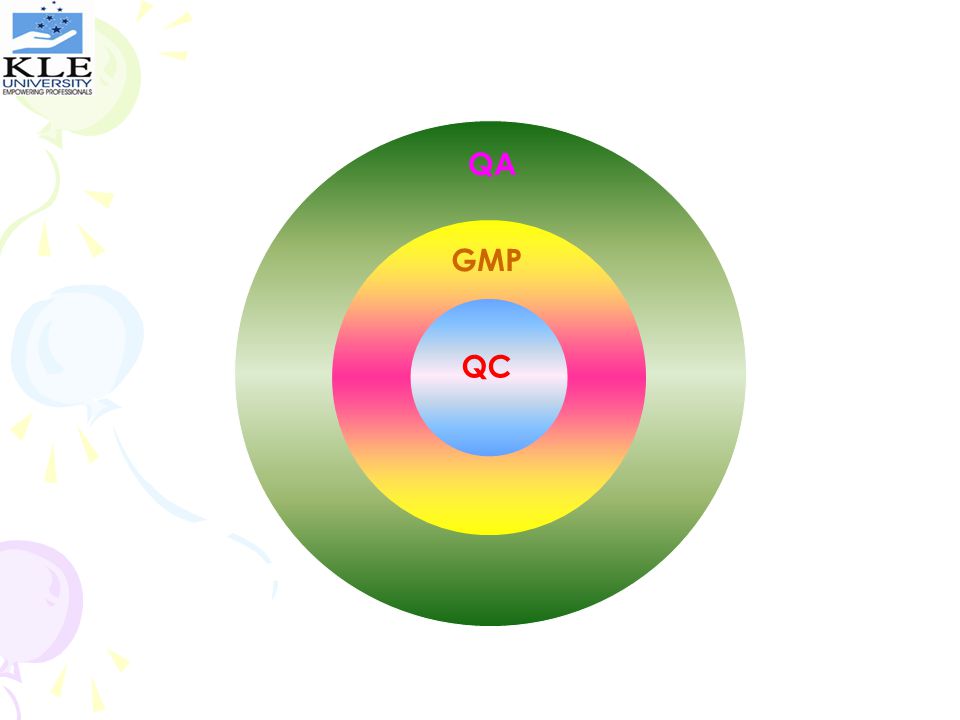
QA GMP QC
3
It is the sum total of the organized arrangements with the objective of ensuring that products will be of the quality required for their intended use QA

4
Is that part of Quality Assurance aimed at ensuring that products are consistently manufactured to a quality appropriate to their intended use GMP

5
Is that part of GMP concerned with sampling, specification & testing, documentation & release procedures which ensure that the necessary & relevant tests are performed & the product is released for use only after ascertaining it’s quality QC

6
QA and QC QC is that part of GMP which is concerned with sampling,
specifications, testing and with in the organization, documentation,and release procedures which ensure that the necessary and relevant tests are carried out QA is the sum total of organized arrangements made with the object of ensuring that product will be of the Quality required by their intended use.

7
QA and QC Operational laboratory techniques and activities used to fulfill the requirement of Quality All those planned or systematic actions necessary to provide adequate confidence that a product will satisfy the requirements for quality

8
QA and QC QA is company based QC is lab based
9
ESTABLISHING ACCEPTANCE CRITERION FOR A SPECIFIED IMPURITY IN A NEW DRUG SUBSTANCE
Determine impurity level in relevant batches1 Is impurity also a degradation product? YES Estimate maximum increase in impurity at retest date using data from relevant accelerated and long-term stability studies Determine mean + upper confidence limit for the impurity (Let this = A) NO Is A or B greater than the qualified level? Acceptance criterion = A or B (as appropriate) NO Determine maximum likely level as: A + increase in degradation product at appropriate storage conditions. (Let this = B) YES 1 Relevant batches are those from development, pilot and scale-up studies. 2 Refer to ICH Guideline on Impurities in New Drug Substances Definition: upper confidence limit = three times the standard deviation of batch analysis data Acceptance criterion = qualified level or establish new qualified level2

10
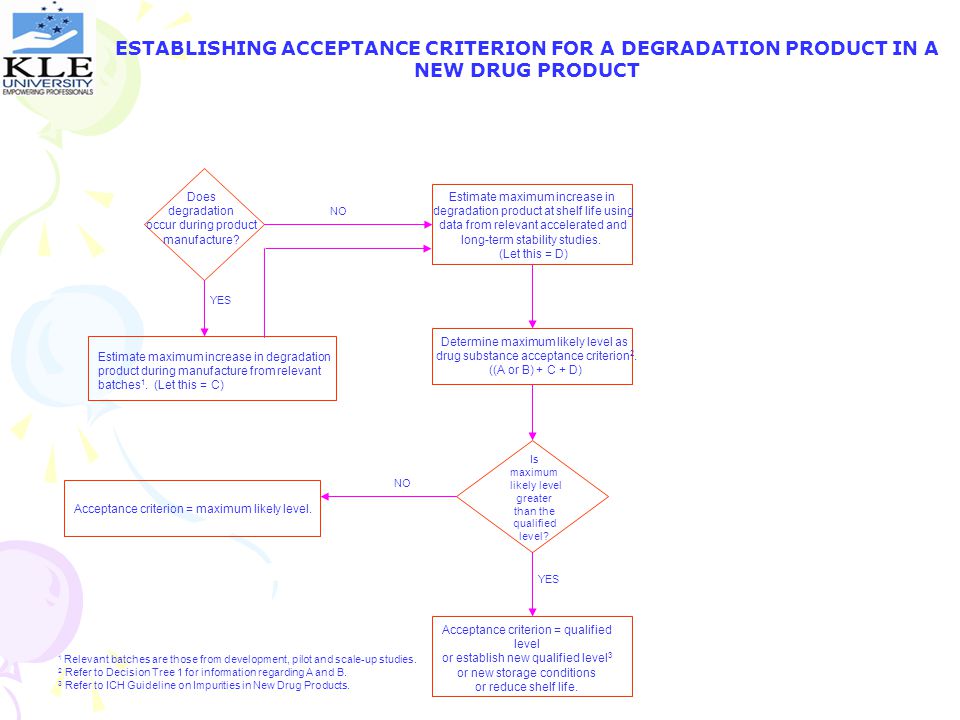
ESTABLISHING ACCEPTANCE CRITERION FOR A DEGRADATION PRODUCT IN A NEW DRUG PRODUCT
Does degradation occur during product manufacture? Estimate maximum increase in degradation product at shelf life using data from relevant accelerated and long-term stability studies. (Let this = D) NO YES Determine maximum likely level as drug substance acceptance criterion2. ((A or B) + C + D) Estimate maximum increase in degradation product during manufacture from relevant batches1. (Let this = C) Is maximum likely level greater than the qualified level? NO Acceptance criterion = maximum likely level. YES Acceptance criterion = qualified level or establish new qualified level3 or new storage conditions or reduce shelf life. 1 Relevant batches are those from development, pilot and scale-up studies. 2 Refer to Decision Tree 1 for information regarding A and B. 3 Refer to ICH Guideline on Impurities in New Drug Products.
11
No Acceptance Criterion Required
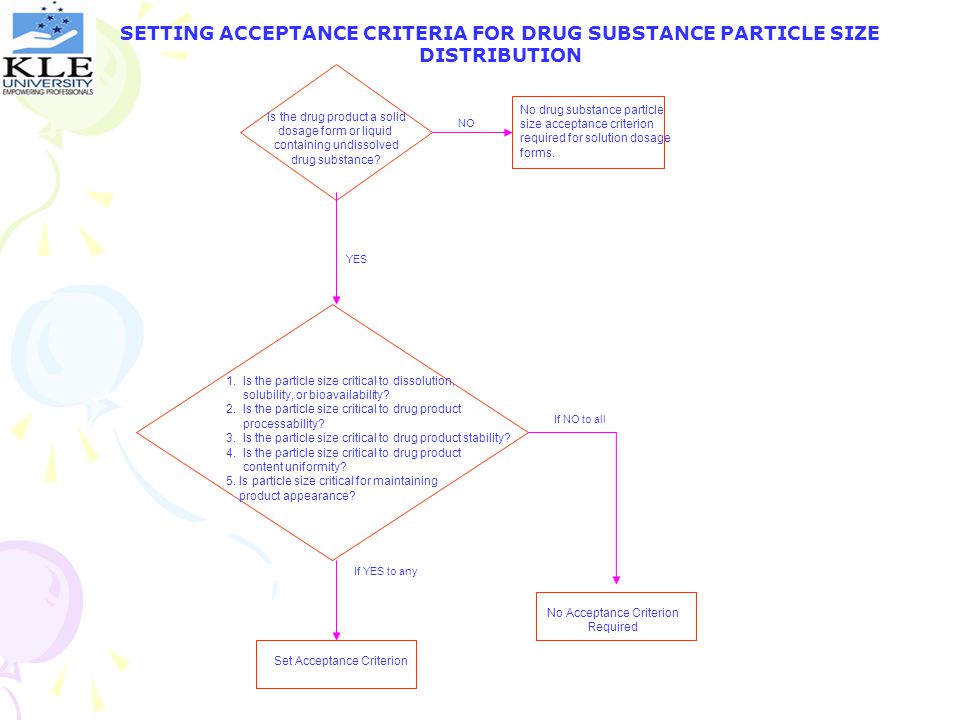
SETTING ACCEPTANCE CRITERIA FOR DRUG SUBSTANCE PARTICLE SIZE DISTRIBUTION No drug substance particle size acceptance criterion required for solution dosage forms. Is the drug product a solid dosage form or liquid containing undissolved drug substance? NO YES 1. Is the particle size critical to dissolution, solubility, or bioavailability? 2. Is the particle size critical to drug product processability? 3. Is the particle size critical to drug product stability? 4. Is the particle size critical to drug product content uniformity? 5. Is particle size critical for maintaining product appearance? If NO to all If YES to any No Acceptance Criterion Required Set Acceptance Criterion
12
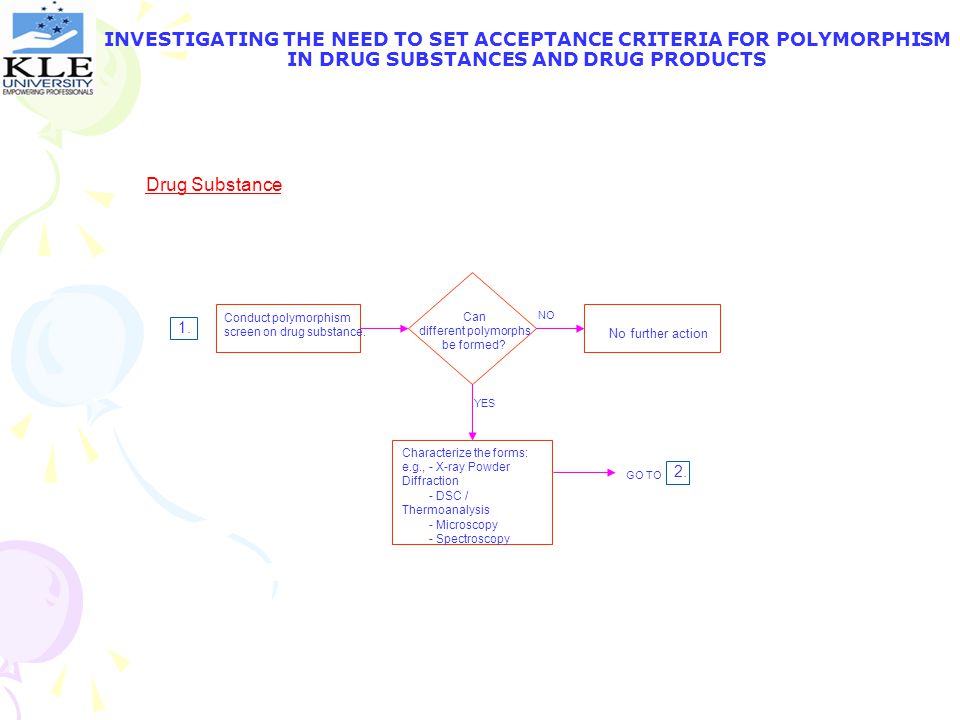
Can different polymorphs be formed?
INVESTIGATING THE NEED TO SET ACCEPTANCE CRITERIA FOR POLYMORPHISM IN DRUG SUBSTANCES AND DRUG PRODUCTS Drug Substance Conduct polymorphism screen on drug substance. Can different polymorphs be formed? NO 1. No further action YES Characterize the forms: e.g., – X-ray Powder Diffraction DSC / Thermoanalysis Microscopy Spectroscopy 2. GO TO
13
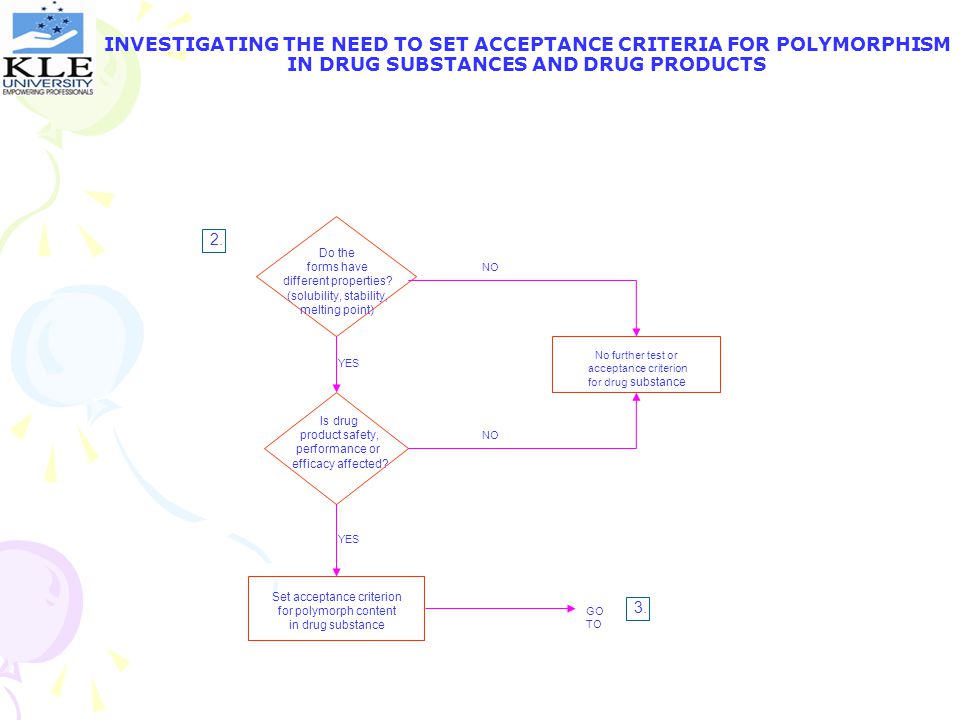
INVESTIGATING THE NEED TO SET ACCEPTANCE CRITERIA FOR POLYMORPHISM IN DRUG SUBSTANCES AND DRUG PRODUCTS 2. Do the forms have different properties? (solubility, stability, melting point) NO No further test or acceptance criterion for drug substance YES Is drug product safety, performance or efficacy affected? NO YES Set acceptance criterion for polymorph content in drug substance 3. GO TO
14
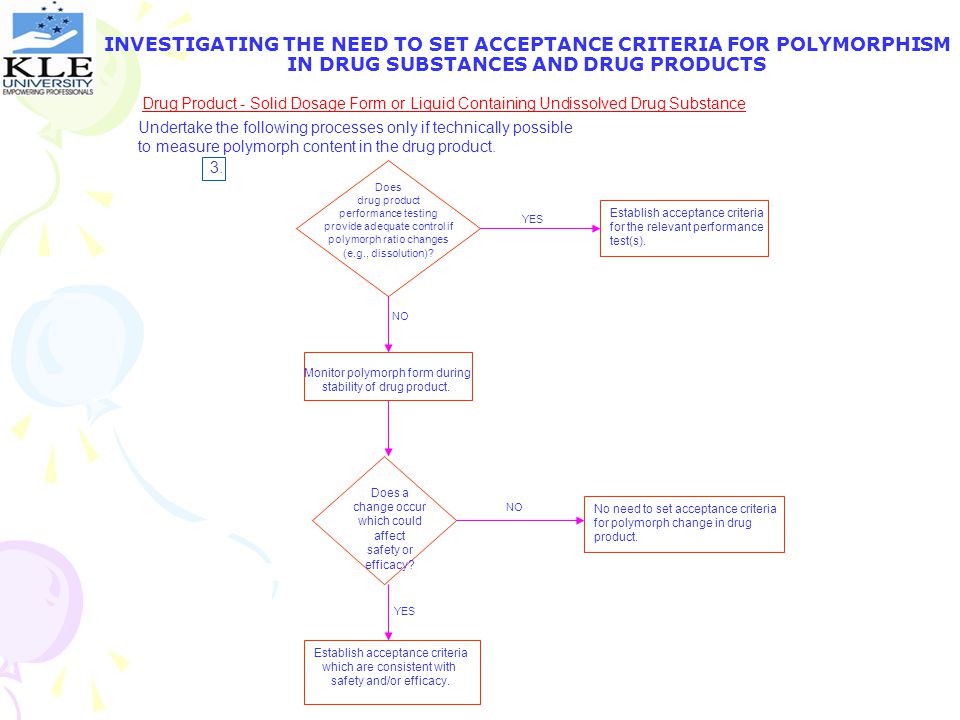
INVESTIGATING THE NEED TO SET ACCEPTANCE CRITERIA FOR POLYMORPHISM IN DRUG SUBSTANCES AND DRUG PRODUCTS Drug Product – Solid Dosage Form or Liquid Containing Undissolved Drug Substance Undertake the following processes only if technically possible to measure polymorph content in the drug product. 3. Does drug product performance testing provide adequate control if polymorph ratio changes (e.g., dissolution)? Establish acceptance criteria for the relevant performance test(s). YES NO Monitor polymorph form during stability of drug product. Does a change occur which could affect safety or efficacy? NO No need to set acceptance criteria for polymorph change in drug product. YES Establish acceptance criteria which are consistent with safety and/or efficacy.
15
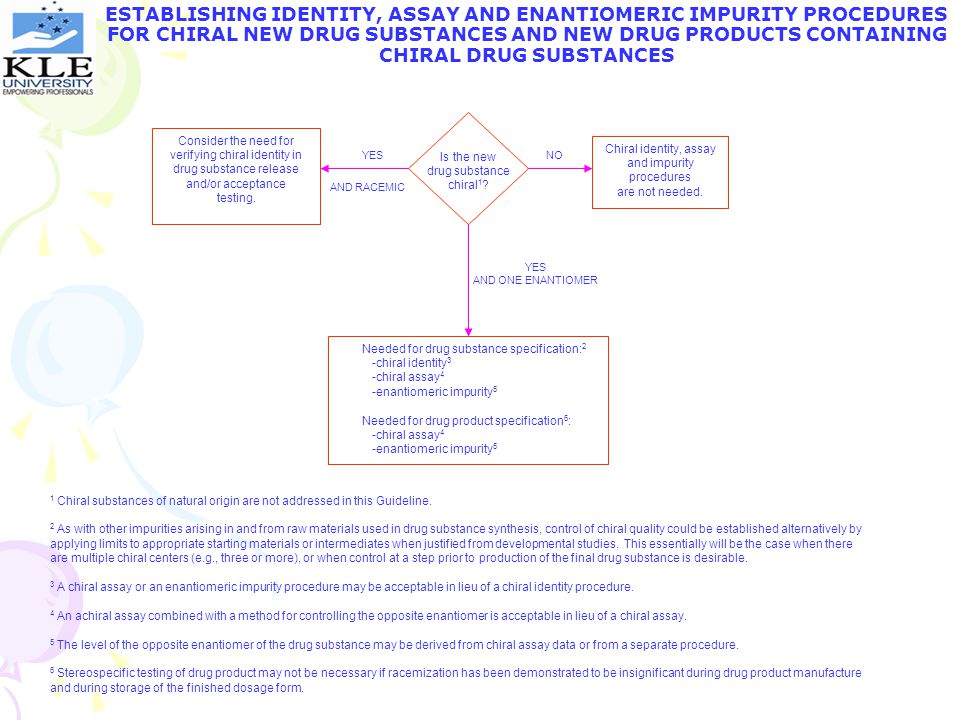
ESTABLISHING IDENTITY, ASSAY AND ENANTIOMERIC IMPURITY PROCEDURES FOR CHIRAL NEW DRUG SUBSTANCES AND NEW DRUG PRODUCTS CONTAINING CHIRAL DRUG SUBSTANCES Consider the need for verifying chiral identity in drug substance release and/or acceptance testing. Chiral identity, assay and impurity procedures are not needed. YES Is the new drug substance chiral1? NO AND RACEMIC YES AND ONE ENANTIOMER Needed for drug substance specification: chiral identity3 -chiral assay4 -enantiomeric impurity5 Needed for drug product specification6: -chiral assay4 -enantiomeric impurity5 1 Chiral substances of natural origin are not addressed in this Guideline. 2 As with other impurities arising in and from raw materials used in drug substance synthesis, control of chiral quality could be established alternatively by applying limits to appropriate starting materials or intermediates when justified from developmental studies. This essentially will be the case when there are multiple chiral centers (e.g., three or more), or when control at a step prior to production of the final drug substance is desirable. 3 A chiral assay or an enantiomeric impurity procedure may be acceptable in lieu of a chiral identity procedure. 4 An achiral assay combined with a method for controlling the opposite enantiomer is acceptable in lieu of a chiral assay. 5 The level of the opposite enantiomer of the drug substance may be derived from chiral assay data or from a separate procedure. 6 Stereospecific testing of drug product may not be necessary if racemization has been demonstrated to be insignificant during drug product manufacture and during storage of the finished dosage form.
16
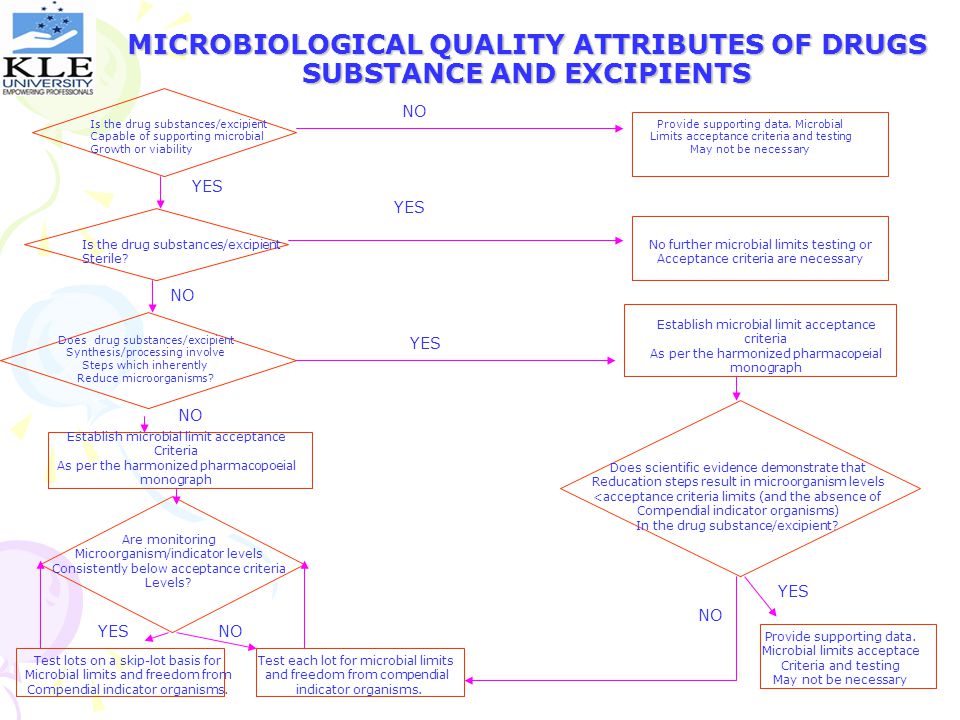
MICROBIOLOGICAL QUALITY ATTRIBUTES OF DRUGS SUBSTANCE AND EXCIPIENTS
NO Is the drug substances/excipient Capable of supporting microbial Growth or viability Provide supporting data. Microbial Limits acceptance criteria and testing May not be necessary YES YES Is the drug substances/excipient Sterile? No further microbial limits testing or Acceptance criteria are necessary NO Establish microbial limit acceptance criteria As per the harmonized pharmacopeial monograph Does drug substances/excipient Synthesis/processing involve Steps which inherently Reduce microorganisms? YES NO Establish microbial limit acceptance Criteria As per the harmonized pharmacopoeial monograph Does scientific evidence demonstrate that Reducation steps result in microorganism levels <acceptance criteria limits (and the absence of Compendial indicator organisms) In the drug substance/excipient? Are monitoring Microorganism/indicator levels Consistently below acceptance criteria Levels? YES NO YES NO Provide supporting data. Microbial limits acceptace Criteria and testing May not be necessary Test lots on a skip-lot basis for Microbial limits and freedom from Compendial indicator organisms. Test each lot for microbial limits and freedom from compendial indicator organisms.
17
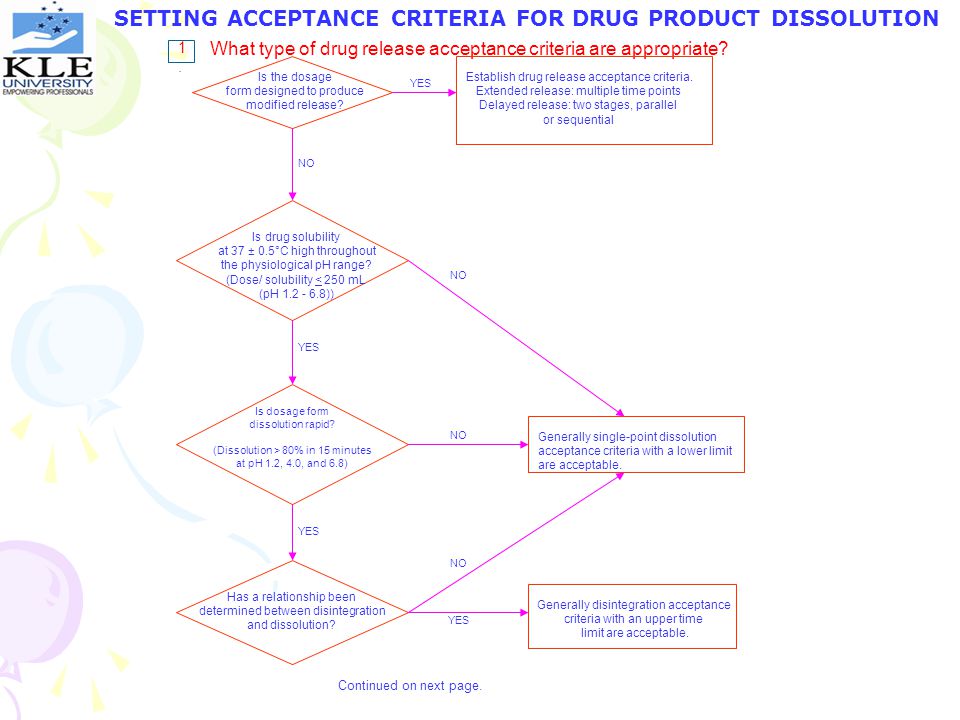
SETTING ACCEPTANCE CRITERIA FOR DRUG PRODUCT DISSOLUTION
1. What type of drug release acceptance criteria are appropriate? Is the dosage form designed to produce modified release? Establish drug release acceptance criteria. Extended release: multiple time points Delayed release: two stages, parallel or sequential YES NO Is drug solubility at 37 ± 0.5°C high throughout the physiological pH range? (Dose/ solubility < 250 mL (pH )) NO YES Is dosage form dissolution rapid? (Dissolution > 80% in 15 minutes at pH 1.2, 4.0, and 6.8) NO Generally single-point dissolution acceptance criteria with a lower limit are acceptable. YES NO Has a relationship been determined between disintegration and dissolution? Generally disintegration acceptance criteria with an upper time limit are acceptable. YES Continued on next page.
18
SETTING ACCEPTANCE CRITERIA FOR DRUG PRODUCT DISSOLUTION
2. What specific test conditions and acceptance criteria are appropriate? [immediate release] Does dissolution significantly affect bioavailability? (e.g., have relevant developmental batches exhibited unacceptable bioavailability?) YES Attempt to develop test conditions and acceptance criteria which can distinguish batches with unacceptable bioavailability. NO Do changes in formulation or manufacturing variables affect dissolution? (Use appropriate ranges. Evaluate dissolution within pH ) YES Are these changes controlled by another procedure and acceptance criterion? YES NO NO Adopt appropriate test conditions and acceptance criteria without regard to discriminating power, to pass clinically acceptable batches. Adopt test conditions and acceptance criteria which can distinguish these changes. Generally, single point acceptance criteria are acceptable.
![2. What specific test conditions and acceptance criteria are appropriate [immediate release] Does dissolution significantly affect bioavailability (e.g., have relevant developmental batches exhibited unacceptable bioavailability ) YES. Attempt to develop test conditions and acceptance. criteria which can distinguish batches. with unacceptable bioavailability. NO. Do changes in formulation or manufacturing variables affect dissolution (Use appropriate ranges. Evaluate dissolution within pH ) YES. Are these changes controlled by another procedure and acceptance criterion YES. NO. NO. Adopt appropriate test conditions and acceptance criteria without. regard to discriminating power, to. pass clinically acceptable batches. Adopt test conditions and acceptance criteria which can distinguish these changes. Generally, single point acceptance criteria are acceptable. SETTING ACCEPTANCE CRITERIA FOR DRUG PRODUCT DISSOLUTION](http://slideplayer.com/slide/5199515/16/images/18/SETTING+ACCEPTANCE+CRITERIA+FOR+DRUG+PRODUCT+DISSOLUTION.jpg)
19
SETTING ACCEPTANCE CRITERIA FOR DRUG PRODUCT DISSOLUTION
3. What are appropriate acceptance ranges? [extended release] Are bioavailability data available for batches with different drug release rates? NO Is drug release independent of in vitro test conditions? YES YES NO Can an in vitro / in vivo relationship be established? (Modify in vitro test conditions if appropriate.) Use all available stability, clinical, and bioavailability data to establish appropriate acceptance ranges. NO YES Use the in vitro / in vivo correlation, along with appropriate batch data, to establish acceptance ranges. Are acceptance ranges >20% of the labeled content? Provide appropriate bioavailability data to validate the acceptance ranges. YES NO Finalize acceptance ranges.
![3. What are appropriate acceptance ranges [extended release] Are bioavailability data available for batches with different drug release rates NO. Is drug release independent of in vitro test conditions YES. YES. NO. Can an in vitro / in vivo relationship be established (Modify in vitro test conditions if appropriate.) Use all available stability, clinical, and bioavailability data to establish appropriate acceptance ranges. NO. YES. Use the in vitro / in vivo correlation, along with appropriate batch data, to establish acceptance ranges. Are acceptance ranges >20% of the labeled content Provide appropriate bioavailability data to validate the acceptance ranges. YES. NO. Finalize acceptance ranges. SETTING ACCEPTANCE CRITERIA FOR DRUG PRODUCT DISSOLUTION](http://slideplayer.com/slide/5199515/16/images/19/SETTING+ACCEPTANCE+CRITERIA+FOR+DRUG+PRODUCT+DISSOLUTION.jpg)
20
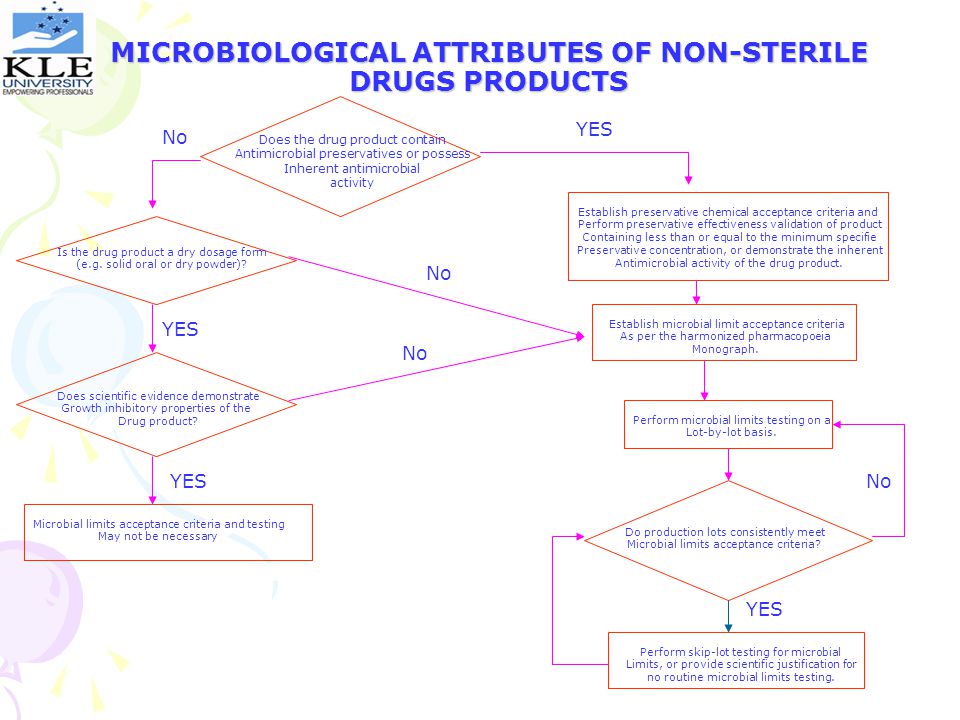
MICROBIOLOGICAL ATTRIBUTES OF NON-STERILE DRUGS PRODUCTS
YES No Does the drug product contain Antimicrobial preservatives or possess Inherent antimicrobial activity Establish preservative chemical acceptance criteria and Perform preservative effectiveness validation of product Containing less than or equal to the minimum specifie Preservative concentration, or demonstrate the inherent Antimicrobial activity of the drug product. Is the drug product a dry dosage form (e.g. solid oral or dry powder)? No YES Establish microbial limit acceptance criteria As per the harmonized pharmacopoeia Monograph. No Does scientific evidence demonstrate Growth inhibitory properties of the Drug product? Perform microbial limits testing on a Lot-by-lot basis. YES No Microbial limits acceptance criteria and testing May not be necessary Do production lots consistently meet Microbial limits acceptance criteria? YES Perform skip-lot testing for microbial Limits, or provide scientific justification for no routine microbial limits testing.
21
ICH Harmonised Tripartite Guideline
Stability Testing of New Drug Substances and Products Stability Testing: Photostability Testing of New Drug Substances and Products Stability Testing for New Dosage Forms Bracketing and Matrixing Designs for Stability Testing of New Drug Substances and Products Evaluation for Stability Data Stability Data Package for Registration Applications in Climatic Zones III and IV Validation of Analytical Procedures: Text and Methodology Impurities In New Drug Substances

22
ICH Harmonised Tripartite Guideline
Impurities in New Drug Products Impurities: Guideline for Residual Solvents Evaluation and Recommendation of Pharmacopoeial Texts for Use in the ICH Regions on Microbiological Examination of Non-Sterile Products: Microbial Enumerations Tests Evaluation and Recommendation of Pharmacopoeial Texts for Use in the ICH Regions on Microbiological Examination of Non-Sterile Products: Test for Specified Micro-Organisms

23
ICH Harmonised Tripartite Guideline
Evaluation and Recommendation of Pharmacopoeial Texts for Use in the ICH Regions on Microbiological Examination of Non-Sterile Products: Acceptance Criteria for Pharmaceutical Preparations and Substances for Pharmaceutical Use Evaluation and Recommendation of Pharmacopoeial Texts for Use in the ICH Regions on Residue on Ignition/Sulphated Ash Evaluation and Recommendation of Pharmacopoeial Texts for Use in the ICH Regions on Test for Extractable Volume of Parenteral Preparations

24
ICH Harmonised Tripartite Guideline
Evaluation and Recommendation of Pharmacopoeial Texts for Use in the ICH Regions on Test for Particulate Contamination: Sub-Visible Particles Evaluation and Recommendation of Pharmacopoeial Texts for Use in the ICH Regions on Disintegration Evaluation and Recommendation of Pharmacopoeial Texts for Use in the ICH Regions on Uniformity of Dosage Units Evaluation and Recommendation of Pharmacopoeial Texts for Use in the ICH Region on Dissolution Test

25
ICH Harmonised Tripartite Guideline
Evaluation and Recommendation of Pharmacopoeial Texts for Use in the ICH Regions on Sterility Test Evaluation and Recommendation of Pharmacopoeial Texts for Use in the ICH Regions onTablet Friability Evaluation and Recommendation of Pharmacopoeial Texts for Use in the ICH Regions on Polyacrylamide Gel Electrophoresis Viral Safety Evaluation of Biotechnology Products Derived from Cell Lines of Human or Animal Origin

26
ICH Harmonised Tripartite Guideline
Quality of Biotechnological Products: Analysis of the Expression Construct in Cells used for Production of r-DNA Derived Protein Products Quality of Biotechnological Products: Stability Testing of Biotechnological/Biological Products Derivation and Characterisation of Cell Substrates Used for Production of Biotechnological/Biological Products Comparability of Biotechnological/Biological Products Subject to Changes in their Manufacturing Process Specifications: Test Procedures and Acceptance Criteria for New Drug Substances and New Drug Products: Chemical Substances

27
ICH Harmonised Tripartite Guideline
Specifications: Test Procedures and Acceptance Criteria for Biotechnological/Biological Products Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients Pharmaceutical Development Quality Risk Management Pharmaceutical Quality System Quality Implementation Working Group

28
Quality Assurance (QA) Management Procedure

29
How to write Standard Operating Procedure
SOP describes standard SOP format that you can use immediately for your quality procedure. SOP has instructions on how to write a formal operating procedure for your systems which your people can follow everyday.

30
All Documents-Classifications, Definition and Approval Matrix
In this SOP you will find all type of quality and Technical/Master file documents to build up a good quality management system for your manufacturing sites, definition of documents, their classification, approval requirements and retention requirements. This procedure has schematic diagrams for your understanding of how different types of documents are prepared and stored in a typical documentation.

31
Quality Documentation Management and Change Control
This SOP describes how to generate new quality documents or change control of existing documents, review of quality documents, satellite file management, role of document author, approver, document control officer and satellite file administrator. In this SOP you will also find numbering systems of different quality documents like audit files, SOP’s, forms, manuals, training files, QA agreements, project files etc and their effective archiving system.

32
Documentation Rule for GMP Documents
This SOP describes the principles to be followed in GMP documents, entry of data and information, signature requirements and correction technique of incorrectly entered data or information.

33
Quality Documentation-Control, Tracking and Distribution
In this SOP you will find mainly the role of document control officer during the initiation, creation, circulation and approval of new quality related documents. It also describes the procedure of modification and review of existing document using a documentation database. Management of existing and superseded documents is also a art of this procedure. You will see all the forms referred during the instruction are attached at the end of the procedure.
![]()
34
Preparation, Maintenance and Change Control of Master Documents
This SOP particularly focused on the management of master file documents like specifications, control methods, raw materials, finished goods and packaging specification and test reports, formulation, stability files etc required to generate during the product registration in the market. This SOP gives instruction on their creation, change control, numbering system, approval requirements and maintenance in a simple master file database. You will see all the forms referred during the instruction are attached at the end of the procedure.

35
Deviation Report System
It is a regulatory requirement to capture all sorts of deviations evolves in your systems in order to maintain the continuous improvement to your processes and systems. This SOP describes how to categorize the deviations between production, audit, quality improvements, technical deviations, customer complaints and environmental, health and safety deviations. It describes the management responsibilities of initiating deviation, capture data, analysis, investigation, determination of assignable causes, generation of management report and initiatives to be taken on corrective and preventative actions.

36
Shelf Life of Product This simple SOP describes the meaning of shelf life and provides on how to interpret shelf lives and storage conditions for your raw materials from the Certificate of Analysis, determining expiry date for your finished products by use of raw material date of manufacturing and their shelf lives.

37
Vendor Selection and Evaluation
This SOP describes the procedure to be followed during the vendor assessment and vendor evaluation for purchasing of raw materials, critical and non critical packaging components, laboratory supplies, engineering supplies and imported finished goods from the vendor. These instructions are essential for approving prospective vendor.

38
Vendor Certification This procedure aim to describe the process by which a vendor may be certified to supply materials or services. This procedure applies to vendors that supply a material or service to be used at any stage of manufacture by operations. Here you will get the roles of each department in the process to certify an approved vendor.

39
Product Complaint Procedure
This procedure covers the receipt, logging, evaluation, investigation and reporting system of all complaints received from customers for the marketed products. This SOP contains step by step instruction to be followed in the customer complaint management like numbering of complaint, registration, evaluation of complaints, determination of assignable cause for the complaint deviation, implementation of corrective and preventive actions, trending of complaints and handling of counterfeit products.

40
Annual Product Review This procedure provides a guideline to annual product review which is required to be performed for each product produced for the commercial market to evaluate data, trends and to identify any preventative or corrective action that would lead to product quality improvements and report them to management.

41
Rework Procedure This SOP contains the step by step instruction to be followed when the rework of an in-process or completed finished good is required. This SOP covers the reworks of in-process manufactured goods where new batch number is introduced for the reworked part and rework of manufactured finished good keeping the same batch number. This SOP also describes how to create rework protocols for each individual case.

42
Authorized Person This simple procedure describes the accreditation, accountabilities and responsibilities of an Authorized person, responsible for release of finished goods for sale.

43
Product Identification and Traceability
The purpose of this SOP is to define the method used for the identification of all contributing materials that could affect product quality and to ensure their full traceability. Here you will find instruction on all the records and documents used for the identification and traceability of incoming raw materials and out going finished goods.

44
Audits This SOP describes the process of planning, performing, reporting and follow-up of different audits for your systems like Internal Quality audit, Vendor audit, Environmental Health and Safety (EHS) audit, EHS workplace inspection, Housekeeping audit. This SOP also describes the process to be followed by manufacturing personnel during an audit from a Regulatory authority.

45
Example-Checklist for Batch Documentation
This SOP describes the identification of all documentation relevant to a production process in the form of “Batch Documentation Checklists” and to ensure their collection by completion of the checklists by Authorized Persons. This procedure is based on an example of tablet packaging process described in the ‘Manufacturing’ category.

46
Evaluation of Batch Documentation and Release for Sale
This procedure describes the process of collection, evaluation and record of batch related document generated during the production of a batch before an authorized person can release the batch for sale. This procedure is based on an example of tablet packaging process described in the ‘Manufacturing’ category.

47
GMP Training This SOP describes how to design and deliver GMP related training for your manufacturing staffs, training assessment design, recording of assessment and preparation of training reports.

48
How to Write Training Materials
This simple SOP contains instructions on how to write training materials, identification of training requirements, available resources, preparation of training aid checklists for your manufacturing staffs.

49
House Keeping Audit Procedure
This SOP describes the requirements, checklists and reporting procedure on housekeeping audits. Individual checklist forms are attached at end of the procedure for different areas like process, laboratory, engineering stores, warehouses. This procedure also describes the handling of non-compliance found during the housekeeping audits.

50
Management and Control of Contract Work
The procedure describes the management and control of contract work provided by the contractors for packaging and finished products for your company as well as control of contract works done by your company on behalf of others.

51
Criteria for Sourcing of RM, Critical Packaging Components and Imported Finishing Goods
The purpose of this SOP is to describe the process for approval of an external vendor/manufacturer supplying products to your company. It covers raw materials (including bulk products for subsidiaries and contract manufacturers), critical packaging components in contact with product and imported finished goods. The SOP also references affiliated documentation detailing the scope of active materials used and the approved manufacturers of these materials.

52
Quality Concern Investigation Process
This procedure contains instruction to be followed when conducting Investigations and to raise and assess Deviation Report when an investigation or incident Investigation occurs. This procedure is to be used in conjunction with SOP, which covers the approval and follow-up activities associated with a Deviation Report. Here you will find collection of information for an incident or a deviation, steps to be followed for a cross functional investigation, reporting and implementing of the outcomes of investigation.

53
Quality Control Laboratory Procedures

54
Retest Dating of Raw Materials
The purpose of this procedure is to describe how to run the expired stock report; to describe how to define the requirements for the retesting and assignment of storage period for active ingredients, excipients and raw materials; to instruct retesting procedure and to determine the status of a finished goods batch with a shorter shelf life.

55
Calibration Policies for Laboratory Instruments
This SOP describes the calibration polices of laboratory instruments/equipments. It describes labeling and security requirements of laboratory instruments/equipments. This SOP also describes the investigational steps to be required in the case of failed calibration

56
Archiving Laboratory Documentation
This procedure describes retention and disposal procedures of laboratory documentation, general laboratory documentation system that includes handling of rejected raw material and finished product reports, finished goods certificate of analysis, finished goods register, raw material certificate of analysis, register, trend cards, procedure for long term document retention.

57
Management of Reference Substances
This SOP describes the ordering referencing, storing, coding, use and general register maintenance of primary and impurity reference substances, primary reagent reference solutions, secondary raw material reference substance, assay testing procedure of secondary raw material reference substance, use of secondary raw material reference substance in the laboratory routine analysis, determination of expiry date and re-test date of reference substances.

58
Laboratory Workbook This SOP describes types of laboratory workbooks, general and GMP requirements of using workbooks, analytical data entry in the workbook, formatting of laboratory workbooks for routine testing, experiments and trials, workbook retention policy, instruction on data entry for incomplete experiments and additional data.

59
Creation of Certificate of Analysis
The purpose of this procedure is to define the content and format of a Certificate of Analysis (C/A) and Certificate of Manufacture (C/M) and to provide guidance for issuing a Certificate of Analysis or Certificate of Manufacture and to locate the appropriate data required for this task.

60
Managing Analytical Reagents
This procedure identifies the need for all analytical reagents and solutions prepared from the reagents, to have an assigned expiry date and storage conditions recorded on the label. Here you will find the procedure for purchase and management of analytical reagents and laboratory prepared reagents.

61
Laboratory Waste Management
This simple procedure describes how to dispose off laboratory generated wastes of toxic, explosive, flammable, corrosive, oxidizing and biologically damaging natures.

62
Retention Samples-Laboratory
The purpose of this SOP is to describe the finished good and raw material sample retention procedures, products manufacture and/or received onsite and/or chemically tested by the laboratory.

63
Laboratory Supplier Approval
In this simple SOP you will find the procedure for approving laboratory suppliers and criteria for the purchase of equipment, instrumentation, consumables, durables and glassware for the laboratory.

64
Laboratory Results-Out of Specification Investigation
This procedure describes the actions to be taken by an analyst in the event the result of a test does not conform to raw material/components or finished products specifications for physical and chemical tests. An out of specification (OOS) result does not necessarily mean the batch under investigation fails and shall be rejected. The OOS result shall be investigated and the findings of the investigation, including re-test results shall be interpreted to evaluate the batch and reach a decision regarding release or rejection.

65
Raw Materials-Laboratory Testing and Documentation
This SOP describes the procedure for sampling, location, pre-testing, testing and documentation of all raw materials and components subject to test, out of specification results, microbiological tests and release procedure for passed raw materials and components.

66
Finished Goods-Laboratory Testing and Documentation
This SOP describes the procedure for sampling, location, pre-testing, testing and documentation of all finished products subject to test, reagents and standards to be used for analysis, management of out of specification results, microbiological tests and release procedure for passed finished goods.

67
Preparation and Maintenance of Stability Protocols (Pharmaceuticals)
This procedure describes the preparation and management of stability protocols for marketed products. This procedure is applicable to all protocols for stability studies on commercial products. The responsibility of the commercial Site stability manager for creating and maintaining protocols that are required for studies that came as a result of validation or process deviation.

68
Stability and Trial Testing Procedure (Pharmaceuticals)
To describes the steps necessary to ensure the effective control of stability and trial testing programs of new and existing products. This procedure is focused on setting up of stability programs, testing, reporting, general sampling procedure for stability programs, data generation and analysis, annual maintenance of stability, new product stability procedure, procedure for in-house trials, reporting and interpretation of trials and conclusion of the trail program.

69
REFERENCES

70
THANK YOU Cell No:







